WHODrug Change Request
Is there a medicinal product name missing from WHODrug? Or do you have questions regarding a pre-existing record in WHODrug? Please do not hesitate to submit a Change Request.
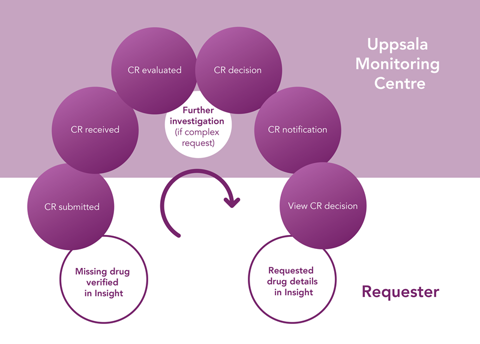
A Change Request (CR) can relate to the inclusion of a new medicinal product in WHODrug or modification of an existing record.
Key features
- Both single- and multiple drug requests in a batch can be submitted as well as proposals for new Standardised Drug Groupings (SDG) and ATC changes.
- Using WHODrug Change Request will make it easier to keep track of submitted requests and to search for previously submitted requests. The requester can also view requests submitted by users from the same company.
- Newly approved drug names will be included in the earliest possible release of WHODrug and drug details can be viewed instantly in WHODrug Insight’s Upcoming data.
When submitting single Change Requests you will normally receive a first response from UMC within three business days. Complex requests and large batch requests may require longer time to process.
Read more about the Change Request process
How to access WHODrug Change Request
WHODrug Change Request is available to users with a valid WHODrug Global subscription and a personal UMC username and password. If you have any questions, do not hesitate to contact us.